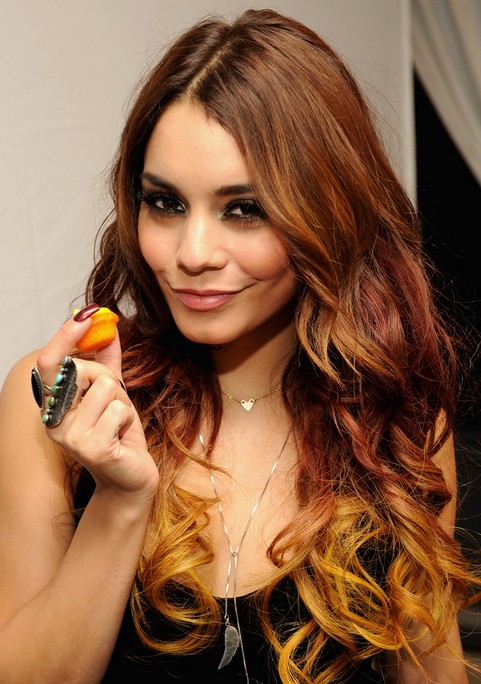One of the most frustrating trends I've come across (not nearly as bad as leggings-as-pants, of course) is ombre hair. And the reason that it frustrates me isn't because it's ugly. It's because ombre hair colouring is typically misunderstood and poorly executed. Ombré,
according to Webster's dictionary, means "shaded or graduated in tone". This to me calls for a subtle gradient between a few similar shades or tones of the same colour. (Not the same as dip-dying!!)
Here is one example of a beautiful ombre look: a natural/gold tone in a blend of different shades from medium to dark blonde. This perfectly mimics the look that natural blondes have in the winter when new hair has grown in but the ends are still sun-bleached from the summer.
This can even be done with less natural colours like the picture below. Unlike the previous, this isn't varying levels in the same colour family; it's varying shades in the same colour level. Vanessa has a copper tone followed by a cherry tone, finished by a yellow tone. The gradient in this look is good, but if a customer asked for this I'd be inclined to go cherry-copper-yellow, because that's the natural gradient you see in a rainbow and it flows a lot better visually.
Photos like those make me happy that ombre has taken dominance as one of the big hair trends right now: after the chunky highlighted and half-and-half styles of the 90s stayed popular through the 2000s without a lot of change, it's a relief to see colouring with better blending and more subtle pops of colour taking over.
But then there's this...unless you use many different shades to do the transition from dark to blonde, you get that look like you bleached your hair out a long time ago and never bothered colouring back. Let's say the dark here is a level 5, and the blonde a 9. Without taking hours with precise detail work blending 5 to 6 to 7 to 8 to 9, the best you could likely do is use a 7 in the middle and 9 on the ends...still not a great look.
But this type of look is worse. This is not ombre, this is dip-dying. If you have a severe line halfway down your hair, no blending or gentle transition has taken place. If your hair looks like a dual fiber paintbrush, please call it dip-dyed and leave poor ombre alone.

So how do you avoid a harsh line of colour? First off, stay close to your own colour. You can venture out by a few shades, or into the realm of different tones of the same shade. But black and blonde will never be ombre...say goodbye to your 1999 zebra highlights. Secondly, use a good ombre painting technique. The easiest way (and what I've done in my own hair, adding a cool red tone to the ends of my natural brown hair) is to put your hair in two high ponytails: this makes your hair very accessible both in proximity to your face, and by allowing access to different layers of your hair (think about how a low nape-of-the-neck ponytail looks smooth, while a high crown ponytail looks layered). Then get some hair colour that you can easily apply with your hands: a mousse formula is ideal. Squirt some of the colour mousse into gloved hands and scrunch into the ponytails like you would regular styling mousse, focusing on the ends. This will give a natural blended look with one colour.
For two or more colours, you may want to use foils. Within a piece of foil, brush on some water or hair conditioner onto your original hair colour starting at the scalp and going 1/3 of the way down the shaft. The water or conditioner will keep other colours from affecting your natural colour. Then paint the middle third of the strand with your first colour, overlapping the conditioner by a few centimetres. Finally use the second colour to paint the lower third of the strand to the ends, again overlapping with the previous colour (this creates the blended transition area).
Good luck and stay ombre!